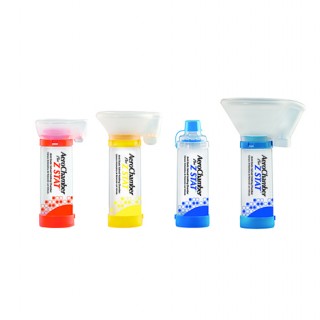
AEROCHAMBER PLUS® Z STAT® AVHC
THE MONAGHAN ADVANTAGE
- The original valved holding chamber design
- Cost effective
- Anti-static, can be used directly out of package
- Increased aerosol suspension time
- Optimized delivery system for consistent medication availability
- Easy to clean – dishwasher safe
- Low resistance valve
- Recommended by leading MDI manufacturers
- Compatible with all commonly prescribed MDIs
- Not Made or Manufactured with BPA, Natural Rubber Latex, Phthalates, Lead, or PVC
- Designed and manufactured in United States except for silicone components
PRODUCT DESCRIPTION
The innovator in valved holding chambers, the AeroChamber® brand of valved holding chamber (aVHC) has been trusted by hospitals for over 30 years. The AeroChamber Plus® Z STAT® aVHC proprietary valve baffle design increases aerosol suspension time to assist dose assurance of metered dose inhaler medication. The optimally designed anti-static chamber ensures consistent medication availability.
The cost effective choice for hospitals, the AeroChamber Plus® Z STAT® aVHC combines patient-centric design features that optimize respirable drug dose. Supported by hundreds of in vitro and in vivo studies, including publication in the New England Journaland CHEST, the AeroChamber Plus® Z STAT® aVHC is also supported by numerous third party clinical evaluations for safety and efficacy.
THE FACTS
- The original valved holding chamber has evolved into the AeroChamber Plus® Z STAT® aVHC to interface with improved MDI aerosol formulations and activities. The AeroChamber Plus® Z STAT® aVHC continues to offer the best value for optimal aerosol drug delivery and economic value for hospitals.
- Different chambers deliver different amounts of medication.1 The AeroChamber Plus® Z STAT® aVHC provides a consistent dose of medication substantially equivalent to the MDI dose.2
- Designed to respond to shallow or low volume breathing patterns, the extra-sensitive inhalation valve on the AeroChamber Plus® Z STAT® aVHC makes it easier for patients to inhale the medication.
Download the AEROCHAMBER PLUS® Z STAT® AVHC
sell sheet.
References
1 Drug Safety Update – Inhaled products that contain corticosteroids, July 2008. Medicines and Healthcare Products Regulatory Agency.
2 TMI Lab Data. Studies on file.